Trusted information about health treatments should be based on evidence. This is high quality research carried out by qualified medical professionals or scientists with no conflicts of interest. Evidence-based health information can help you make vital decisions about your care.
How can I tell which clinical evidence is best?
There are many types of clinical evidence. The pyramid of evidence below was developed by doctors to help them rate the quality of evidence. Generally, the more people included in research, the more reliable the findings.
Why would lower quality evidence be used?
Guidelines and reviews do not exist for all topics. So, evidence from lower down the pyramid will be used. Case reviews of small groups of people are viewed as lower quality evidence. But they can be useful, for example, in rare disease.
How can I be sure about the evidence?
Research should be checked by other experts before it is published. This is called peer review. Health information should tell you if the evidence is uncertain or if medical experts have different opinions.
What does research tell me about my treatment?
Everyone is different. Research can tell you the most likely outcome. It cannot guarantee what will happen to you but it can help you make an informed choice.
What about ‘real world’ patient data?
Real world data is collected outside of clinical trials. It can be come from sources like electronic health records. This is important because some groups of people are under-represented in clinical trials. This data is now included in systematic reviews and clinical guidelines.
Can I share my data?
You can help build evidence on your condition or treatment by sharing your data. You may be asked to give consent to your healthcare team or researchers. Check how your data will be used and how your identity will be protected.
Pyramid of evidence
The pyramid of evidence ranks clinical evidence from high to low quality. The best evidence is at the top. These are clinical guidelines and systematic reviews. Experts check all the clinical research and patient data available and use a set system to make recommendations based on the findings.
Clinical trials research new medicines. They are also called randomised controlled or placebo studies. One group of patients is given a new treatment and a similar group is given a traditional treatment or a placebo. A placebo is a dummy treatment with no medical value. Researchers compare what happens to both groups.
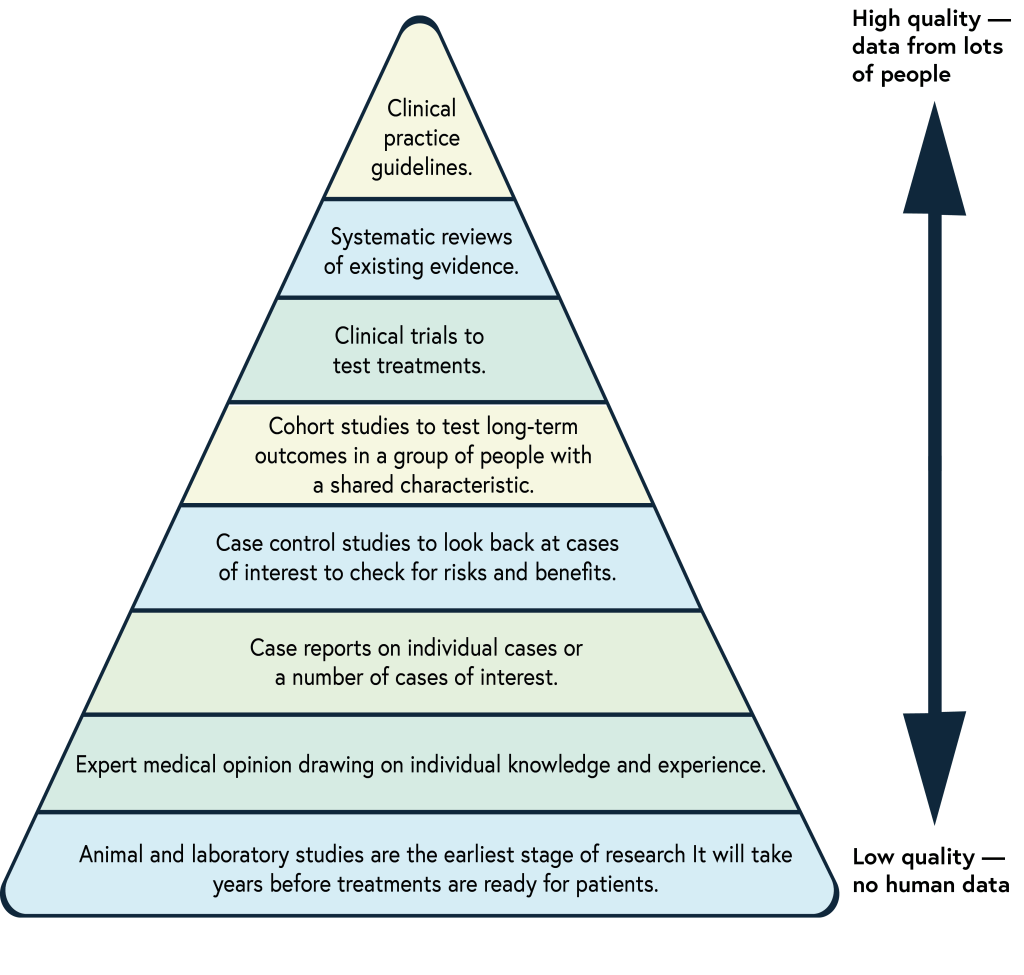
Adapted from The Evidence Based Medicine Pyramid.


